Atomization of CO2 Extracts
Atomization occurs when a jet of liquid is broken down into more or less fine droplets. The ideal spray consists of drops of the same diameter. There are different technologies to obtain a micronized powder with CO2. The best known are: NEA (Near-critical Expansion Atomization), RESS (Rapid Expansion of Supercritical Solution), PGSS (Particles from Gas Saturated Solutions), SAS (Supercritical Anti Solvent) and SAA (Supercritical Assisted Atomization).
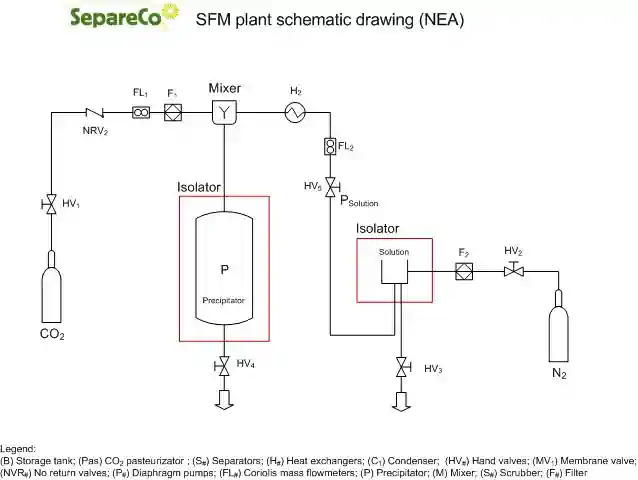
In NEA (Near-critical Expansion Atomization) process, the carbon dioxide is used in this process for the atomization and the crystallization of the product. The product subjected to the process is maintained in the liquid phase in a feed tank at a controlled temperature and subsequently conveyed, at the desired pressure, to the “atomization tower” where there is contact with the carbon dioxide released to atmospheric pressure.
The product obtained can be immediately inserted into the stages of production, mixed or packaged in a modified atmosphere using the carbon dioxide derived from the process. With NEA process special applications are possible, for example in the pharmaceutical field, where fine particulates are required (5 – 30 micron) for the insertion of active ingredients in a matrix.

RESS (Rapid Expansion of Supercritical Solution) is used generally to produce fine particles for the food, cosmetic and pharmaceutical industry.
- Dissolving the solid substance in a SCF,
- Formation of particles due to supersaturation.
In the RESS process, at first SCF-CO2 is pumped at desired pressure and temperature to extraction chamber containing solid substance(s) through heat exchanger. The supercritical solution expands rapidly in the precipitator causing a collapse of pressure and temperature, which favors the formation of drops and particles.
The SCF percolates and dissolves the solid substance(s) in the extractor and then the resulted solution is depressurized through a heated nozzle or capillary at supersonic speed into a low pressure chamber. The supercritical solution is expanded adiabatically in the chamber, which leads to a rapid drop in temperature and pressure and spontaneous formation of droplets/particles. During the rapid expansion of the supercritical solution, the density and solvent power decrease significantly, resulting in super saturation of the solution and consequently precipitation of desire particles free of a residual solvent. The RESS process advantages are its simplicity, effectiveness and the absence of organic solvents.
There is also a variation of the traditional RESS that uses a solid cosolvent (RESS-SC). This concept is widely used in the field of extraction with SCF and is called co-extraction. Generally, the RESS process is batch type, but there is also a continuous RESS process, which allows to obtain micro-particles in a constant and continuous way.
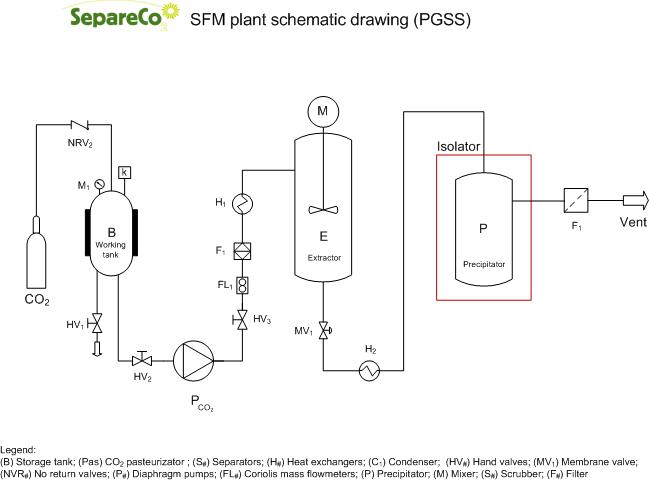
PGSS (Particles from Gas Saturated Solutions) is a technique for the production of microparticles of different materials of relatively low melting temperatures, such as polymers, waxes or fats. The process is based on the capacity of those materials to dissolve large amounts of CO2 at moderate pressures. Upon depressurization down to ambient conditions, the dissolved CO2 is rapidly released and expanded, producing an intense cooling effect that promotes the formation of microparticles.
The PGSS process is quite similar to the RESS process with one important difference: in PGSS process, the polymer(s) are first melted or suspended in solvent at a given temperature in an autoclave and then solubilizing SCF-CO2 in above melted or liquid suspended substance(s), leading to a so called gas saturated solution or suspension that is further depressurized through a nozzle with the formation of droplets or solid particles.
Unlike to RESS technique, the principle governing PGSS process involves both the pressure and temperature and solvent-induced phase separation. This technique avoids the low solubility in SC-CO2 of many molecules of pharmaceutical interest such as proteins and peptides that would be too difficult to treat with RESS.
Advantages of PGSS process are:
- substance need not be soluble in SCF-CO2,
- simplicity of this process, leading to low processing cost and wide range of application,
- can be used with suspensions of active ingredient(s) in polymer(s) or other carrier substance leading to composite particles,
- can be applied to process inorganic powders to pharmaceutical compounds,
- low solvent gas usage and pressure than RESS process as operational condition.
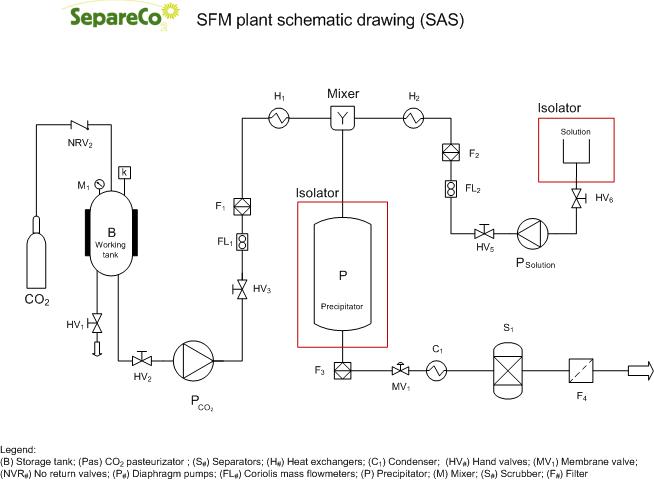
As mentioned previously, the low solubility of a part of pharmaceutical products of interest limits the use of SC-CO2 as a solvent in the mico or nano production of particels. To solve this problem it was decided to use the SC-CO2 as anti-solvent and not as solvent. In this case the solute is insoluble in the anti-solvent, while the anti-solvent must be miscible with the liquid solvent. The process is based on a quite simple concept: when a liquid solution is sufficiently expanded by a gas, the liquid phase is no longer a good solvent for the solute causing the precipitation with formation of particles.
SAS (Supercritical Anti Solvent), according to its name, applies the supercritical fluid as an antisolvent. Hence the solute to be micronized has to be quasi non-soluble in the supercritical fluid. This process is structured in a differently way than previous RESS and PGSS. The SCF is first pumped to the top of the high pressure vessel until the system reaches a constant temperature and pressure. Subsequently, active substance solution is sprayed as fine droplets into above SCF bulk phase through an atomization nozzle. The large volume expansion of drug solution in vessel, resulting dissolution of SCF into liquid droplets and, subsequently, in super saturation due to reduction in solvent power leading to nucleation and formation of small and mono disperse particles.
Particles are collected on a filter at the bottom of the vessel. The SCF and organic solvent mixture flow down to a depressurized tank where suitable temperature and pressure condition allow gas-liquid separation. After the collection of sufficient quantity of particles, the spraying of liquid solution has to be stopped. Furthermore, to remove residual solvent, pure SCF continues to flow through the vessel.
There are many variations of this process:
- ASAIS (Atomization of Supercritical Antisolvent Induces Suspension). In ASAIS process, antisolvent induced precipitation occurs in a small tube, where antisolvent mixed with the solution to generate a suspension. This suspension of particles is then sprayed into a precipitator at atmospheric condition for solvent separation, which eliminates the high volume and high pressure precipitator. In addition, very small to moderate antisolvent concentration is required. Contrary to SAS process, the particles recovery is performed by cyclone separator rather than using filter.
- SEDS (Solution Enhanced Dispersion by Supercritical fluids). This is a modification of SAS process in which the SCF and drug solution are introduced simultaneously in to the precipitation vessel at particular temperature and pressure through the coaxial nozzle. The design of co-axial nozzle is such that to facilitate the dispersion of drug solution by SCF, thereby enhancing mass transfer and formation of fine particles. In addition, the high velocity of SCF allows intense mixing with drug solution. Here, the SCF serves both as an antisolvent and as a dispersion medium.
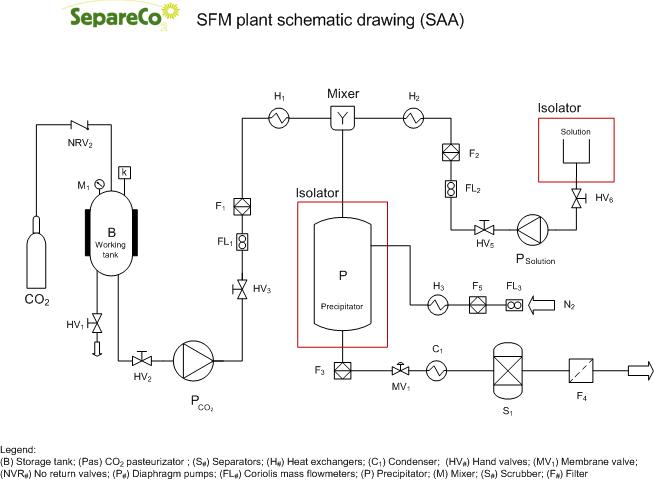
SAA (Supercritical Assisted Atomization) process is focused on the nebulization of the liquid solution rather than using dense gas (SCF) to achieve precipitation by solubility reduction for the solute to be micro- or nano-sized. At first, the solute is dissolved or suspended in aqueous or organic solvent or their mixture and then mixed intimately with near critical or SC by pumping both fluid through a near zero volume tee to generate an emulsion. The resultant emulsion is rapidly expanded through a flow restrictor to near atmospheric pressure to form aerosol consisting of micro droplets and micro bubbles. The aerosol is formed due to sudden dispersion of the liquid solution caused by rapid expansion of compressed gas. The drying chamber is filled with heated air or nitrogen gas to maintain the desired temperature for rapid drying of aerosol droplets or micro bubbles. Dry particles are collected on a filter placed at the outlet of the drying chamber.
Parameters influencing the particle formation are flow rate of solution percentage of dissolved or suspended substance, inner diameter flow restrictor (50-175 μm), temperature of the drying chamber, residence time of droplets or micro bubbles (as micro bubbles are dried faster than droplets). This process is also known as CAN-DB (Carbon dioxide Assisted Nebulization with Bubble Dryer). The SAA process differs for the use of a saturator to enable a better mixing of the supercritical fluid with the solute containing the product before it is injected into the precipitator. Generally the saturator is made with fillings for generating a large exchange surface. Others use the principle of cavitation to achieve the same result.


